7:53 AM Chemical reaction |
 Chemical reactionChemical reactions such as combustion in the fire, fermentation and the reduction of ores to metals were known since antiquity. Initial theories of transformation of materials were developed by Greek philosophers, such as the Four-Element Theory of Empedocles stating that any substance is composed of the four basic elements – fire, water, air and earth. In the Middle Ages, chemical transformations were studied by Alchemist. They attempted, in particular, to convert lead into gold, for which purpose they used reactions of lead and lead-copper alloys with sulfur.The production of chemical substances that do not normally occur in nature has long been tried, such as the synthesis of sulfuric and nitric acids attributed to the controversial alchemist Jābir ibn Hayyān. The process involved heating of sulfate and nitrate minerals such as copper sulfate, alum and saltpeter. In the 17th century, Johann Rudolph Glauber produced hydrochloric acid and sodium sulfate by reacting sulfuric acid and sodium chloride. With the development of the lead chamber processLeblanc process, allowing large-scale production of sulfuric acid and sodium carbonate, respectively, chemical reactions became implemented into the industry. Further optimization of sulfuric acid technology resulted in the contact process in 1880s, and the Haber process was developed in 1909–1910 for ammonia synthesis in 1746 and the From the 16th century, researchers including Jan Baptist van Helmont, Robert BoyleIsaac Newton tried to establish theories of the experimentally observed chemical transformations. The phlogiston theory was proposed in 1667 by Johann Joachim Becher. It postulated the existence of a fire-like element called "phlogiston", which was contained within combustible bodies and released during combustion. This proved to be false in 1785 by Antoine Lavoisier who found the correct explanation of the combustion as reaction with oxygen from the air.and Joseph Louis Gay-Lussac recognized in 1808 that gases always react in a certain relationship with each other. Based on this idea and the atomic theory of John Dalton, Joseph Proust had developed the law of definite proportions, which later resulted in the concepts of stoichiometry and chemical equations. Regarding the organic chemistry, it was long believed that compounds obtained from living organisms were too complex to be obtained synthetically. According to the concept of vitalism, organic matter was endowed with a "vital force" and distinguished from inorganic materials. This separation was ended however by the synthesis of ureaFriedrich Wöhler in 1828. Other chemists who brought major contributions to organic chemistry include Alexander William Williamson with his synthesis of ethers and Christopher Kelk Ingold, who, among many discoveries, established the mechanisms of substitution reactions. from inorganic precursors by EquationsChemical equations are used to graphically illustrate chemical reactions. They consist of chemical or structural formulas
of the reactants on the left and those of the products on the right.
They are separated by an arrow (→) which indicates the direction and
type of the reaction. The tip of the arrow points in the direction in
which the reaction proceeds. A double arrow ( More complex reactions are represented by reaction schemes, which in addition to starting materials and products show important intermediates or transition states. Also, some relatively minor additions to the reaction can be indicated above the reaction arrow; examples of such additions are water, heat, illumination, a catalyst, etc. Similarly, some minor products can be placed below the arrow, often with a minus sign.
Elementary reactionsThe elementary reaction is the smallest division into which a chemical reaction can be decomposed to, it has no intermediate products. Most experimentally observed reactions are built up from many elementary reactions that occur in parallel or sequentially. The actual sequence of the individual elementary reactions is known as reaction mechanism. An elementary reaction involves a few molecules, usually one or two, because of the low probability for several molecules to meet at a certain time.
In a typical dissociation reaction, a bond in a molecule splits resulting in two molecular fragments. The splitting can be homolytic or heterolytic. In the first case, the bond is divided so that each product retains an electron and becomes a neutral radical. In the second case, both electrons of the chemical bond remain with one of the products, resulting in charged ions. Dissociation plays an important role in triggering chain reactions, such as hydrogen-oxygen or polymerization reactions.
For bimolecular reactions, two molecules collide and react with each other. Their merger is called chemical synthesis or an addition reaction. Another possibility is that only a portion of one molecule is transferred to the other molecule. This type of reaction occurs, for example, in redox and acid-base reactions. In redox reactions, the transferred particle is an electron, whereas in acid-base reactions it is a proton. This type of reaction is also called metathesis. for example
Chemical equilibriumMost chemical reactions are reversible, that is they can and do run in both directions. The forward and reverse reactions are competing with each other and differ in reaction rates. These rates depend on the concentration and therefore change with time of the reaction: the reverse rate gradually increases and becomes equal to the rate of the forward reaction, establishing the so-called chemical equilibrium. The time to reach equilibrium depends on such parameters as temperature, pressure and the materials involved, and is determined by the minimum free energy. In equilibrium, the Gibbs free energy must be zero. The pressure dependence can be explained with the Le Chatelier's principle. For example, an increase in pressure due to decreasing volume causes the reaction to shift to the side with the fewer moles of gas. The reaction yield stabilized at equilibrium, but can be increased by removing the product from the reaction mixture or increasing temperature or pressure. Change in the initial concentrations of the substances does not affect the equilibrium. ThermodynamicsChemical reactions are largely determined by the laws of thermodynamics. Reactions can proceed by themselves if they are exergonic, that is if they release energy. The associated free energy of the reaction is composed of two different thermodynamic quantities, enthalpy and entropy:
Reactions can be exothermic, where ΔH is negative and energy is released. Typical examples of exothermic reactions are precipitation and crystallization, in which ordered solids are formed from disordered gaseous or liquid phases. In contrast, in endothermic reactions, heat is consumed from the environment. This can occur by increasing the entropy of the system, often through the formation of gaseous reaction products, which have high entropy. Since the entropy increases with temperature, many endothermic reactions preferably take place at high temperatures. On the contrary, many exothermic reactions such as crystallization occur at low temperatures. Changes in temperature can sometimes reverse the direction of a reaction, as in the Boudouard reaction: This reaction between carbon dioxide and carbon to form carbon monoxide is endothermic at temperatures above approximately 800 °C and is exothermic below this temperature. Reactions can also be characterized with the internal energy which takes into account changes in the entropy, volume and chemical potential. The latter depends, among other things, on the activities of the involved substances.
] KineticsThe speed at which a reactions takes place is studied by reaction kinetics. The rate depends on various parameters, such as:
Several theories allow calculating the reaction rates at the molecular level. This field is referred to as reaction dynamics. The rate v of a first-order reaction, which could be disintegration of a substance A, is given by: Its integration yields: Here k is first-order rate constant having dimension 1/time, [A](t) is concentration at a time t and [A]0 is the initial concentration. The rate of a first-order reaction depends only on the concentration and the properties of the involved substance, and the reaction itself can be described with the characteristic half-life. More than one time constant is needed when describing reactions of higher order. The temperature dependence of the rate constant usually follows the Arrhenius equation: where Ea is the activation energy and kB is the Boltzmann constant. One of the simplest models of reaction rate is the collision theory. More realistic models are tailored to a specific problem and include the transition state theory, the calculation of the potential energy surface, the Marcus theory and the Rice–Ramsperger–Kassel–Marcus (RRKM) theory. |
|
|
| Total comments: 1 | ||||||
| ||||||

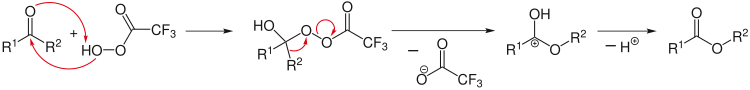
 The most important elementary reactions are unimolecular and
bimolecular reactions. Only one molecule is involved in a unimolecular
reaction; it is transformed by an isomerization or a dissociation
in one or more other molecules. Such reaction requires addition of
energy in the form of heat or light. A typical example of a
unimolecular reaction is the cis–trans
The most important elementary reactions are unimolecular and
bimolecular reactions. Only one molecule is involved in a unimolecular
reaction; it is transformed by an isomerization or a dissociation
in one or more other molecules. Such reaction requires addition of
energy in the form of heat or light. A typical example of a
unimolecular reaction is the cis–trans




![v= -\frac {d[\mathrm{A}]}{dt}= k \cdot [\mathrm{A}]](http://upload.wikimedia.org/math/3/e/a/3eae6de39c4daaaf476d31bb6132f099.png)
![\mathrm{[A]}(t) = \mathrm{[A]}_{0} \cdot e^{-k\cdot t}](http://upload.wikimedia.org/math/1/b/d/1bd6bc5f63af06b606a86fc6654fe682.png)


